Newsletter
The TEI Newsletter
Number 15, March 24, 2006
By: Timothy Parker
 Gamma radiography got its start in the U.S in 1929 at the Naval Research Laboratory for the express purpose of testing thick steel castings. The x-ray machines at that time were very weak, only able to penetrate three inches of steel. As discussed in newsletter #7, March 22, 2005, using radium as a source made it possible to radiograph castings up to 12" thick. The use of this natural source was a tremendous breakthrough, greatly expanding the uses of industrial radiography. However, with a typical energy level of one tenth of one curie for a radium source, exposure times of up to four days were not uncommon. Gamma radiography got its start in the U.S in 1929 at the Naval Research Laboratory for the express purpose of testing thick steel castings. The x-ray machines at that time were very weak, only able to penetrate three inches of steel. As discussed in newsletter #7, March 22, 2005, using radium as a source made it possible to radiograph castings up to 12" thick. The use of this natural source was a tremendous breakthrough, greatly expanding the uses of industrial radiography. However, with a typical energy level of one tenth of one curie for a radium source, exposure times of up to four days were not uncommon.
At the same time, the methods of performing radiography were undergoing change. The figures on this page demonstrate the method known as fishpole radiography. Compared to the techniques we employ today, industrial radiography in the early part of the 20th century was a pretty scary proposition.
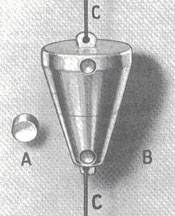
In 1940, radiography sources looked like this and usually contained about one-tenth of a curie of radium. The radium was sealed in a capsule (A) and placed in an aluminum-alloy container (B). The source was handled by using the cord (C) attached the container.
 The technology gains made during World War II were driven in large part by the Navy' s shipbuilding program, and by 1946, the introduction of manmade sources such as cobalt and iridium had all but replaced radium. As this discipline expanded, so did scientific and engineering boundaries. The ability to determine internal soundness in a non-destructive manner reduced the cost and increased the reliability and performance of all types of building materials. The technology gains made during World War II were driven in large part by the Navy' s shipbuilding program, and by 1946, the introduction of manmade sources such as cobalt and iridium had all but replaced radium. As this discipline expanded, so did scientific and engineering boundaries. The ability to determine internal soundness in a non-destructive manner reduced the cost and increased the reliability and performance of all types of building materials.
This picture of fishpole radiography was probably taken during the 1940s. The photo was obviously staged. The sources then available were so weak that exposures of an hour or more were necessary. Even the steadiest hand could not be still long enough to avoid a blurry radiograph.
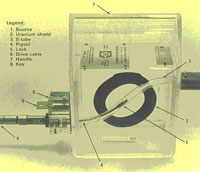
TEI has a number of different sources of radiation, their use depending on the application. X-ray machines produce radioactivity only when they are energized, and the energy level can be adjusted. Sources are radioactive isotopes that emit gamma radiation all the time. As the energy level of these sources cannot be adjusted, exposure times are varied depending on the density and thickness of the material.
 Typically, x-ray machines produce cleaner radiation, and clearer pictures, and our machine is limited to about 130 kV. Low energy levels (50 kV) are used for very thin sections of most metals, moderate thicknesses of graphite and beryllium, small electronic components, wood, plastics, and other common materials. Energy in the 100 kV range is used for 1 aluminum or the equivalent, and higher energy (150 kV) is used for 5 aluminum or 1 of steel. While our machine can be used in the field, it is cumbersome, and the power requirements are demanding. Typically, x-ray machines produce cleaner radiation, and clearer pictures, and our machine is limited to about 130 kV. Low energy levels (50 kV) are used for very thin sections of most metals, moderate thicknesses of graphite and beryllium, small electronic components, wood, plastics, and other common materials. Energy in the 100 kV range is used for 1 aluminum or the equivalent, and higher energy (150 kV) is used for 5 aluminum or 1 of steel. While our machine can be used in the field, it is cumbersome, and the power requirements are demanding.
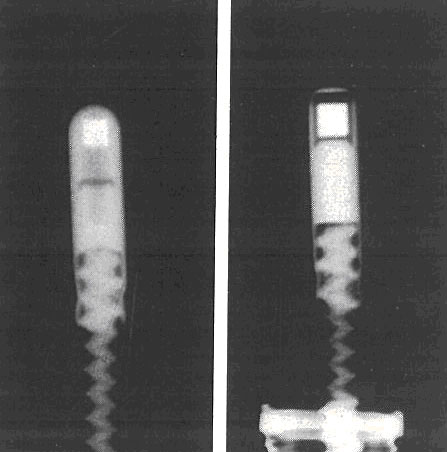
A radiograph of two radiography sources shows the radioactive material inside the steel capsules. The white squares are iridium ¨C 192. The capsules are attached to steel cables.
Sources emit radiation naturally and continuously, and cannot be adjusted in their intensity. A new iridium 192 source has an energy level corresponding to 600kV, and a new cobalt 60 source has an energy level of 1.3MeV. With these intensities, three inches of steel or 16 of concrete can be studied (in the case of iridium), and up to 10 of steel, using cobalt. Sources are much more portable, and are capable of deeper penetrations into materials.
 The benefits of source radiation are tempered by two drawbacks. The first is the cleanliness of the radiation source radiation might best be described as wild. Though the higher energy levels give greater penetration through materials, it is at the expense of clarity. Additionally, radioactive isotopes have a limited useful lifespan, based on what is known as their half-life. Their natural radioactivity occurs when atoms decay from unstable high-energy particles into stable, non-radioactive atoms, giving off radiation as the by-product. The half-life is the time it takes for a source to decay to half its former strength. Iridium has a half-life of 74 days, and cobalt has a half-life of 5.3 years. In contrast, plutonium, element 94, has a half-life of more than 24,000 years! The strength of a source has commonly been measured in curies, or Ci, which corresponds to 3.7 x 1010 disintegrations per second. The SI units for source strength are becquerels. One becquerel, or Bq, is one disintegration per second. The benefits of source radiation are tempered by two drawbacks. The first is the cleanliness of the radiation source radiation might best be described as wild. Though the higher energy levels give greater penetration through materials, it is at the expense of clarity. Additionally, radioactive isotopes have a limited useful lifespan, based on what is known as their half-life. Their natural radioactivity occurs when atoms decay from unstable high-energy particles into stable, non-radioactive atoms, giving off radiation as the by-product. The half-life is the time it takes for a source to decay to half its former strength. Iridium has a half-life of 74 days, and cobalt has a half-life of 5.3 years. In contrast, plutonium, element 94, has a half-life of more than 24,000 years! The strength of a source has commonly been measured in curies, or Ci, which corresponds to 3.7 x 1010 disintegrations per second. The SI units for source strength are becquerels. One becquerel, or Bq, is one disintegration per second.
 Radiography is one of the most versatile test methods used in modern industry. Employing highly penetrating x-rays or gamma rays that do not damage the part itself, radiography provides a permanent visible film record of internal conditions, containing the basic information by which the soundness can be determined. Today digital images can also be recorded and viewed on a computer. TEI ability to utilize different sources of radiation has given us the flexibility to inspect a broad spectrum of materials. They range from brazed Beryllium components for Space Defense Systems (Star Wars) to concrete structures such as buildings and bridges to fire-damaged electronic components. Radiography is one of the most versatile test methods used in modern industry. Employing highly penetrating x-rays or gamma rays that do not damage the part itself, radiography provides a permanent visible film record of internal conditions, containing the basic information by which the soundness can be determined. Today digital images can also be recorded and viewed on a computer. TEI ability to utilize different sources of radiation has given us the flexibility to inspect a broad spectrum of materials. They range from brazed Beryllium components for Space Defense Systems (Star Wars) to concrete structures such as buildings and bridges to fire-damaged electronic components.
|


 Gamma radiography got its start in the U.S in 1929 at the Naval Research Laboratory for the express purpose of testing thick steel castings. The x-ray machines at that time were very weak, only able to penetrate three inches of steel. As discussed in newsletter #7, March 22, 2005, using radium as a source made it possible to radiograph castings up to 12" thick. The use of this natural source was a tremendous breakthrough, greatly expanding the uses of industrial radiography. However, with a typical energy level of one tenth of one curie for a radium source, exposure times of up to four days were not uncommon.
Gamma radiography got its start in the U.S in 1929 at the Naval Research Laboratory for the express purpose of testing thick steel castings. The x-ray machines at that time were very weak, only able to penetrate three inches of steel. As discussed in newsletter #7, March 22, 2005, using radium as a source made it possible to radiograph castings up to 12" thick. The use of this natural source was a tremendous breakthrough, greatly expanding the uses of industrial radiography. However, with a typical energy level of one tenth of one curie for a radium source, exposure times of up to four days were not uncommon. The technology gains made during World War II were driven in large part by the Navy' s shipbuilding program, and by 1946, the introduction of manmade sources such as cobalt and iridium had all but replaced radium. As this discipline expanded, so did scientific and engineering boundaries. The ability to determine internal soundness in a non-destructive manner reduced the cost and increased the reliability and performance of all types of building materials.
The technology gains made during World War II were driven in large part by the Navy' s shipbuilding program, and by 1946, the introduction of manmade sources such as cobalt and iridium had all but replaced radium. As this discipline expanded, so did scientific and engineering boundaries. The ability to determine internal soundness in a non-destructive manner reduced the cost and increased the reliability and performance of all types of building materials.  Typically, x-ray machines produce cleaner radiation, and clearer pictures, and our machine is limited to about 130 kV. Low energy levels (50 kV) are used for very thin sections of most metals, moderate thicknesses of graphite and beryllium, small electronic components, wood, plastics, and other common materials. Energy in the 100 kV range is used for 1 aluminum or the equivalent, and higher energy (150 kV) is used for 5 aluminum or 1 of steel. While our machine can be used in the field, it is cumbersome, and the power requirements are demanding.
Typically, x-ray machines produce cleaner radiation, and clearer pictures, and our machine is limited to about 130 kV. Low energy levels (50 kV) are used for very thin sections of most metals, moderate thicknesses of graphite and beryllium, small electronic components, wood, plastics, and other common materials. Energy in the 100 kV range is used for 1 aluminum or the equivalent, and higher energy (150 kV) is used for 5 aluminum or 1 of steel. While our machine can be used in the field, it is cumbersome, and the power requirements are demanding. The benefits of source radiation are tempered by two drawbacks. The first is the cleanliness of the radiation source radiation might best be described as wild. Though the higher energy levels give greater penetration through materials, it is at the expense of clarity. Additionally, radioactive isotopes have a limited useful lifespan, based on what is known as their half-life. Their natural radioactivity occurs when atoms decay from unstable high-energy particles into stable, non-radioactive atoms, giving off radiation as the by-product. The half-life is the time it takes for a source to decay to half its former strength. Iridium has a half-life of 74 days, and cobalt has a half-life of 5.3 years. In contrast, plutonium, element 94, has a half-life of more than 24,000 years! The strength of a source has commonly been measured in curies, or Ci, which corresponds to 3.7 x 1010 disintegrations per second. The SI units for source strength are becquerels. One becquerel, or Bq, is one disintegration per second.
The benefits of source radiation are tempered by two drawbacks. The first is the cleanliness of the radiation source radiation might best be described as wild. Though the higher energy levels give greater penetration through materials, it is at the expense of clarity. Additionally, radioactive isotopes have a limited useful lifespan, based on what is known as their half-life. Their natural radioactivity occurs when atoms decay from unstable high-energy particles into stable, non-radioactive atoms, giving off radiation as the by-product. The half-life is the time it takes for a source to decay to half its former strength. Iridium has a half-life of 74 days, and cobalt has a half-life of 5.3 years. In contrast, plutonium, element 94, has a half-life of more than 24,000 years! The strength of a source has commonly been measured in curies, or Ci, which corresponds to 3.7 x 1010 disintegrations per second. The SI units for source strength are becquerels. One becquerel, or Bq, is one disintegration per second. Radiography is one of the most versatile test methods used in modern industry. Employing highly penetrating x-rays or gamma rays that do not damage the part itself, radiography provides a permanent visible film record of internal conditions, containing the basic information by which the soundness can be determined. Today digital images can also be recorded and viewed on a computer. TEI ability to utilize different sources of radiation has given us the flexibility to inspect a broad spectrum of materials. They range from brazed Beryllium components for Space Defense Systems (Star Wars) to concrete structures such as buildings and bridges to fire-damaged electronic components.
Radiography is one of the most versatile test methods used in modern industry. Employing highly penetrating x-rays or gamma rays that do not damage the part itself, radiography provides a permanent visible film record of internal conditions, containing the basic information by which the soundness can be determined. Today digital images can also be recorded and viewed on a computer. TEI ability to utilize different sources of radiation has given us the flexibility to inspect a broad spectrum of materials. They range from brazed Beryllium components for Space Defense Systems (Star Wars) to concrete structures such as buildings and bridges to fire-damaged electronic components.